
Also, FDA removes OAI status for Watson's Florida facility, executive management changes as GSK, more...

Also, FDA removes OAI status for Watson's Florida facility, executive management changes as GSK, more...
Scientists are giving up on a preventive vaccine for AIDS, but there are lessons to be learned.

Getting IT, engineering, and manufacturing on the same page requires a delicate balance.

Enterprise process control and management (EPCAM) is a new strategy for healthcare manufacturers based on recent process-control breakthroughs in the electronics industry.

ISA 100.11a and WirelessHART both seek to become the global standard for industrial wireless automation.

Editors' Picks of Pharmaceutical Science & Technology Innovations
Brief pharmaceutical news items for May 2008.

The adoption of computers into every aspect of the pharmaceutical industry has been enormous. Some estimates would put productivity and efficiency gains across the pharmaceutical industry at 2–3 times what they were previously. However, they are only just beginning to have an effect in the areas of training and document management, which have traditionally been managed by people and paper systems. With increasingly strict regulations and the growing complexity of processes and training requirements, there are even greater efficiency gains, as well as cost savings, to be achieved by adopting electronic document management systems (eDMS), learning management systems (LMS) and systems for the electronic signing of documents.

Also, Genzyme to build R&D center in Beijing, Noven Pharmaceuticals appoints Peter C. Brandt president and CEO, more...

Also, GSK expands in Ireland, executive appointments at ProGenTech, more...

Also, Alcon plans to open Singapore facility, Pharmacopeia president and CEO retired, more...

Also, Jubilant Organosys to acquire DRAXIS Health, PPD's Paul Covington to retire, more...

The deadline for the implementation of California's prescription drug tracking system, ePedigree, has been delayed until January 2011.

One more step closer to finalization, the ISA 100 Wireless Compliance Institute presented a live demonstration of a prototype of the ISA 1001.11a wireless standard-based products at the 2008 Wireless Conference in Chongquing, China.

Also, VaxGen and Raven terminate merger agreement, Darren Head appointed CEO of Cytovance, more...

The FDA itself issues a cry for help. Is anybody listening?
Manufacturers expect to see the latest developments in process equipment at INTERPHEX, and this year's show was no disappointment. Exhibitors regularly display additions to their lines of encapsulators, tablet presses, material-handling machines, and other automated manufacturing equipment. But the products on view in Philadelphia Mar. 26–28 were not limited to manufacturing applications.
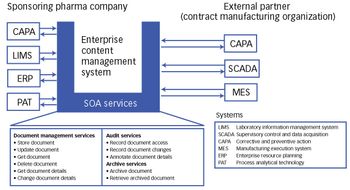
As a skipping stone creates ripples in a lake, SOA can help create benefits that quickly ripple through many other areas of the organization and partners.

Also, Alkermes announces restructuring and reduction of workforce, Icagen announces several senior management promotions, more...

Also, Pipex Pharmaceuticals implements cost-cutting measures, Pfizer's Senior Vice-President and General Counsel Allen Waxman leaves the company, more...

Packaging processes, like other pharmaceutical operations, benefit from the speed and repeatability that automation brings. Robotics in particular provide flexibility and accuracy. In some packaging applications such as carton loading, robotics also perform more efficiently than dedicated machines.

This year, INTERPHEX will take place in Philadelphia, Pennsylvania, on March 26?28. Equipment and Processing Report asked RJ Palermo, industry vice-president of Reed Life Sciences and INTERPHEX, to share his thoughts and expectations about the upcoming event.

Also, PDL BioPharma will no longer pursue sale of the company, executives resign from Topigen Pharmaceuticals, more...

Also, Millipore plans to open Singapore facility, Michael J. Simms joins Alexza Pharmaceuticals, more...

Show blasts off this month in Philadelphia with more suppliers, new trends, and real-world solutions.