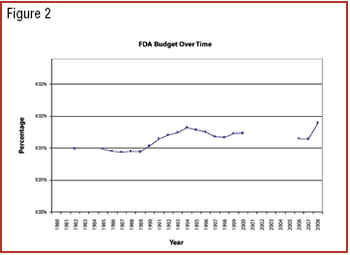
Rep. John D. Dingell (D-MI), chairman of the US House of Representatives Committee on Energy and Commerce, and Rep. Bart Stupak (D-MI), chairman of the Oversight and Investigations Subcommittee, sent letters to the US Food and Drug Administration, Shaw Science Partners, and EthicAd to request information about a new FDA website.