
Emerging pharmaceutical companies represent an important client base for CROs and CMOs. Lessons learned for successful customer–supplier relations.

Emerging pharmaceutical companies represent an important client base for CROs and CMOs. Lessons learned for successful customer–supplier relations.
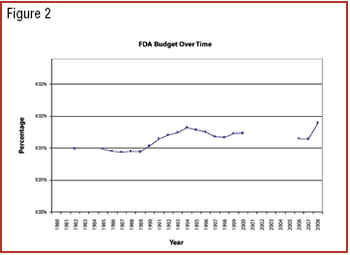
Can previous trends of Democratic and Republican administrations predict industry's future?

Also, Alpharma advises shareholders to reject King's offer; ImClone rejects raised BMS offer; Immunogen appoints Daniel M. Junius, more...
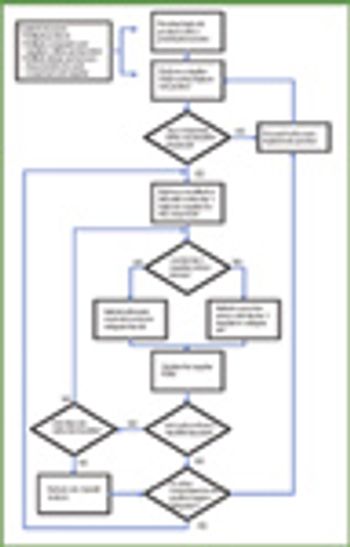
The growth and globalization of the pharmaceutical supply chain make risk assessment more important than ever for pharmaceutical manufacturers. The authors describe a program to identify, prioritize, mitigate, and communicate risks in manufacturer–supplier relationships.
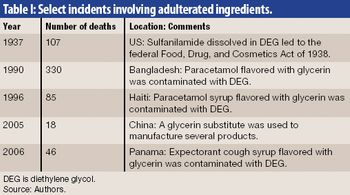
Securing the integrity of the excipient supply chain is a crucial task in ensuring the overall pharmaceutical supply chain. The authors outline excipient-control strategies and practices for the manufacture, distribution, and receipt of excipients.

SAFC, Regis Technologies, Carbogen Amcis, Cambridge Major Laboratories, and AMRI are among the companies at CPhI Worldwide in Frankfurt this week that are moving forward with expansions.

Ionic liquids provide an alternative to certain solvents in select reactions used to synthesize intermediates and active pharmaceutical ingredients.

The potent nature of HPAPIs means there must be careful evaluation of the compound for its level of toxicity when considering manufacture.

Also, Zentiva accepts Sanofi's increased takeover bid, Oriel Therapeutics appoints Richard Fuller CEO, more...

The biopharmaceutical company ImClone Systems rejected Bristol-Myers Squibb's previously announced bid of $60 per share in cash or $4.5 billion for acquiring ImClone, citing the bid as "inadequate."

Also, Quintiles to expand Singapore operations; Christine A. Poon, chairman of Johnson & Johnson's pharmaceuticals group, to retire; more...

The European Chemical Industry Council and five national chemical associations representing France, Germany, Italy, Spain, and the United Kingdom, have launched ReachLink. ReachLink is a company founded to help companies participate in the Substance Information Exchange Forum (SIEF), which is designed as an information-sharing vehicle to facilitate companies in meeting requirements under REACH

Also, Human Genome Sciences enters pact with Hospira, Zosano Pharma names Gail Schulze chair and CEO, more...

Boehringer Ingelheim and and Hisoar (Taizhou, China), a pharmaceutical production company, formed a strategic production alliance in China.

Also, Novartis stops development on "Aurograb," Zealand Pharma appoints David H. Solomon as CEO, more...

The pharmaceutical majors deploy green-chemistry strategies to improve the synthesis of active pharmaceutical ingredients and intermediates.

A recent book shows the links between synthesis, catalysis, and environmental protection.

This year, the employment survey will acknowledge the industry's best employers.
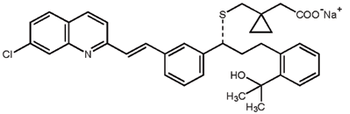
Carbon–hydrogen functionalization, ketone α-alkylation, and biocatalysis are some recent advances in asymmetric synthesis.

Are hypersanitation trends a result of scaremongering or a lack of faith in medicine?

The author explains the planning, equipment, and facility design requried for manufacturing HPAPIs and specialized requirements for handling these compounds.

The outlook is fairly optimistic as contract manufacturing organizations (CMOs) gather at CPhI Worldwide in Frankfurt. CMOs are expanding capacity for small-molecules, biologics, and finished-product manufacturing.
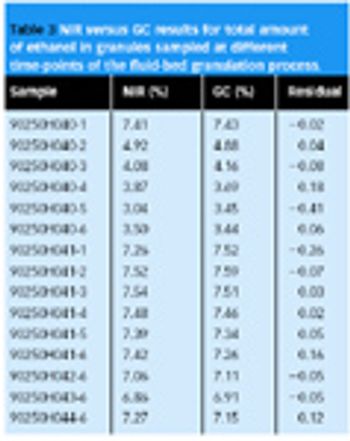
Near-infrared spectroscopy (NIR) is suitable for the analysis of pharmaceutical samples in various solid forms, and can be used for determining chemical properties (e.g., content of drug, water), as well as physical properties (e.g., particle size, tablet hardness).
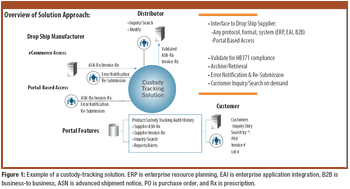
EPedigree, track-and-trace technologies, and other tools for optimizing supply-chain management are of increasing importance to the pharmaceutical industry. The author examines the current regulatory and legislative framework for ePedigree for finished drug products as well as proposals to require electronic statements for pharmaceutical ingredients.

Recombinant microbial whole-cell biocatalysis is a valuable approach for producing enantiomerically pure intermediates. The authors examine several groups of enzymes using this approach: dehydrogenases, hydantoinases, and acylases.