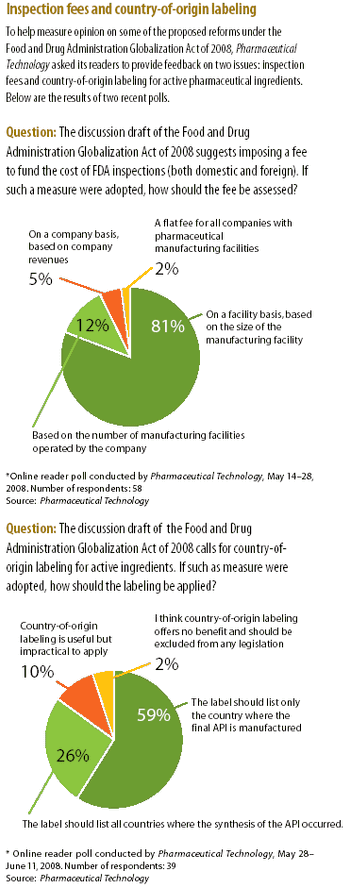
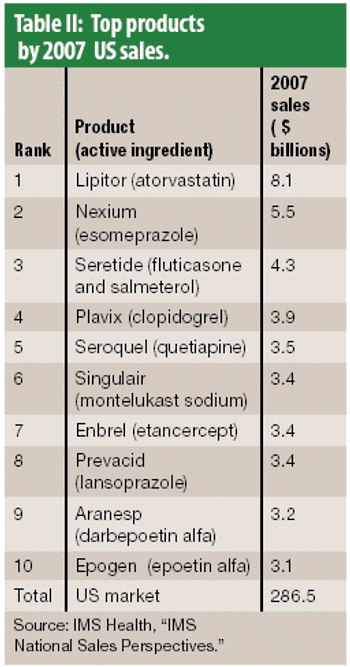
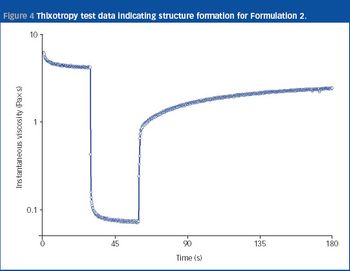
Nasal drug delivery depends on many factors, including the conditions of use by the patient, the drug formulation, and the spray pump and aerosol characteristics. In recent years, the types of drug administered via the nasal route have expanded from locally acting drugs, such as those for allergic rhinitis, to delicate molecules for systemic activity, such as vaccines, proteins and peptides, which can be difficult to administer noninvasively. While the nasal cavity provides a delivery pathway for these large molecules, the rate of mucociliary clearance in the nasal cavity may hinder the extent of absorption. Therefore, formulators must develop mechanisms that improve absorption for high molecular weight compounds.