Advances in engineered particles and the subsequent reduction in the API mass required to achieve a therapeutic dose can lead to a reduction in side effects.

Advances in engineered particles and the subsequent reduction in the API mass required to achieve a therapeutic dose can lead to a reduction in side effects.
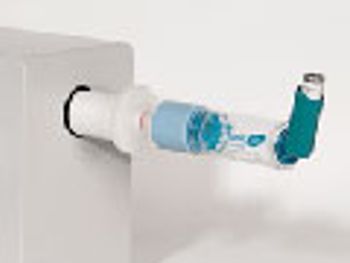
The role of add-on devices and how they affect drug delivery with a pressurized metered dose inhaler.

Biopharma manufacturers must reduce risk in their complex supply chains and establish effective monitoring and control systems.

An expert discusses how companies should be preparing to meet the requirements of the Drug Supply Chain Security Act, which requires phase-in of requirements to prevent counterfeiting.

Manufacturers can identify and reduce waste using tools such as value-stream mapping.

PharmaChk, a portable device being developed to detect substandard medicines, receives $2-million grant.

Biopharma manufacturers must reduce the risk in their complex supply chains

Annual study shows CMO technical expertise is not enough.

Shrinking facility size, growth of biologics, and emerging market demand influence pharma investments.

A techno-economic profiling method supports pharmaceutical process development by helping identify the best manufacturing approach.

The US federal Drug Quality and Security Act (DQSA) and more specifically the Drug Supply Chain Security Act (DSCSA), referring to Title II of the law, requires phase-in of requirements to prevent counterfeiting. An expert discusses how companies should be preparing to meet the requirements of the DSCSA.

Pharmaceutical manufacturers will be-and are already&mdashfacing enormous challenges to implement the serialization regulations within the given timeframe.

Single-use technologies, which started off as a tool for small-volume solution storage and transport has now become the central, enabling technology around which manufacturing processes and facilities are being designed.

In the past 10 years, safety issues have become increasingly important in pharmaceutical packaging, from needle sticking safety to counterfeit protection.

Single-use technologies have gained broad acceptance in biomanufacturing within the past few years, supporting flexible and cost-effective clinical production.

Hovione announced that its API plant in Cork, Ireland has successfully passed a pre-approval inspection by FDA.

New chemical entities with poor aqueous solubility require the use of technologies to enable sufficient oral bioavailability of these NCEs following administration.

Film coating for solid oral dosage forms provide important patient benefits while enhancing brand recognition and adding value to innovator, generic, over-the-counter and nutritional products.

The success of new biopharmaceutical drugs also relies on innovative solutions such as single-use technologies that suppliers have to offer to the pharmaceutical industry.

Innovative therapeutic approaches beyond mAbs will require new methods for future facility construction.

Tool coatings are now seen as an acceptable means of solving production problems, such as sticking and corrosion, resulting in increased product yield.

Single use has introduced a level of flexibility into facilities that was either unheard of or very expensive in a stainless-steel environment.

Quality by Design has initiated a paradigm shift in solid dose pharmaceutical manufacturing.

Efforts to tackle challenges in tablet manufacture are shaped by quality by design (QbD) and continuous manufacturing.

Continuous manufacturing is key to the future of pharmaceutical manufacturing.