
A new class of nanoparticles hold promise for preventing premature drug release and offering greater accuracy and effectiveness in drug delivery.

A new class of nanoparticles hold promise for preventing premature drug release and offering greater accuracy and effectiveness in drug delivery.

Debottlenecking downstream mAb purification.

A technical forum featuring Catalent Pharma Solutions, SAFC, and Neuland Laboratories.

Does nanotechnology offer a cure-all or a kill all? We speak with Ruth Duncan about the real potential of nanomedicines.

We bring industry experts together to discuss the importance of self-administration and what injection technologies are best suited to this cause.

The authors designed an upper punch with a removable punch tip to determine a tablet formulation's propensity to stick by weighing the mass of powder adhered to the punch tip.

Challenges remain, particularly for early-stage biopharmaceutical companies.

Some recent advances involve strategies for accelerating reaction discovery, approaches for inducing chirality and stereochemical analysis, and applications in nanotechnology for protein elucidation.
Industry experts discuss formulation and technical challenges in multilayer tablet manufacture.

Nanosized systems are important in drug delivery. Such nanosized systems include liposomes, nanocrystals, micelles, colloidal particles, quantum dots, and dendrimers. Dendrimers are class of synthetic macromolecules with highly branched, monodispersed, circular, and symmetrical architecture that are used as carrier molecules in drug delivery.
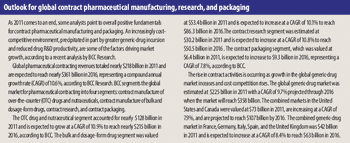
Expansion activity was limited as fine-chemical producers and CMOs of API and intermediates grapple with changing industry fundamentals.

As the excipient supply chain becomes more complex, industry must up the ante to comply with new standards and regulations.

New product reviews for December 2011, focusing on manufacturing.

Drug shortages, supply-chain security, generic-drug incursion, and flexible manufacturing models are some of the issues shaping the bio/pharma industry.

Precedents set in the historic Barr case continue to raise questions over suitable sample-size criteria.

A Q&A with Deborah Tanner, executive vice-president and group president of R&D laboratories at Covance, on recent industry trends.

Readers react to the economic turmoil of the past year and look longingly forward to 2012.

Pharma companies must balance demand for new drugs while facing reduced R&D spending.

The last in a series of eight case studies from the Product Quality Research Institute focuses on internal GMP audits.
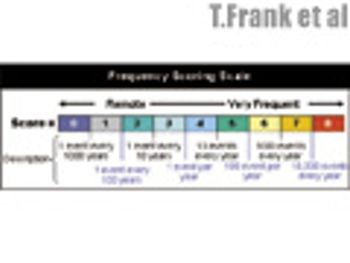
The sixth in a series of eight case studies from the Product Quality Research Institute focuses on packaging line GMP optimization.

On Sept. 27, 2011, FDA sent Genentech a Form 483 listing several violations at the company's South San Francisco, California, plant. The violations included problems with investigations into batch failures, inappropriate equipment design, and insufficient protection against contamination. FDA visited the plant, which produces the cancer drug Avastin, 13 times in September 2011 and made four observations.

Small drug companies with hopes of achieving $1 billion in sales can pursue various strategies.
Pharmaceutical companies are responding to the high cost of introducing new drugs to market in different ways.

Researchers develop various catalytic approaches for improving yield, purity, stereoselectivity, and process conditions.

Novartis AG and Novartis Pharmaceuticals Corporation have been focusing research efforts on rare diseases since the company was established in 1996.