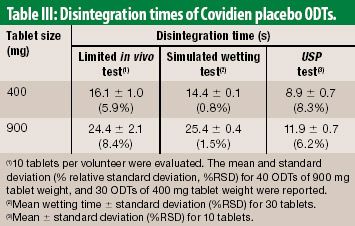
The European Medicines Agency recommended strengthening the warnings for oral moxifloxacin medicines and concluded that these drugs should only be prescribed to treat acute bacterial sinusitis, acute exacerbation of chronic bronchitis, and community-acquired pneumonia when other antibiotics cannot be used or have failed.