
Washington, DC (Feb. 14)?A congressional team has reintroduced the ?Access to Life-Saving Medicine Act,? which establishes a process through which the US Food and Drug Administration can approve generic copies of biologic drugs.

Washington, DC (Feb. 14)?A congressional team has reintroduced the ?Access to Life-Saving Medicine Act,? which establishes a process through which the US Food and Drug Administration can approve generic copies of biologic drugs.

Washington, DC (Jan. 26)-The US Department of Health and Human Services has granted targeted liability protections for manufacturers of a vaccine to prevent a pandemic of influenza A (H5N1).
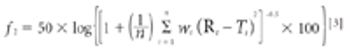
The objectives of this study were to prepare and characterize inclusion complexes of lovastatin with hydroxypropyl-β-cyclodextrin (HPβ-CD) and to study the effect of the complexes on the dissolution rate of lovastatin (LVS). The findings suggest that LVS's poor dissolution profile can be overcome by preparing its inclusion complex with HPβ-CD.
For solid oral-dosage forms, water testing usually is performed to control the chemical, physical, or microbiological properties of the drug product. Measurements of total water as made with Karl Fischer (KF) techniques is not needed and water-activity often will provide a better correlation with changes in chemical, physical, or microbiological properties than KF techniques. In these cases, water activity testing can easily replace KF testing. Water-activity measurements are nondestructive, require little labor, and the equipment required is generally inexpensive. Only a few simple checks are needed to ensure the validity of measurements. Strategies for implementing water activity testing are described.

The same phenomena that create lightning and thunderstorms are around us every day, producing incredibly high voltages, which cause sparks and shocks. Static electricity is a mighty force. Each year excessive electrical charge build cause explosions in the grain industry.1 Look around any flammable storage area and you will see both grounding bars on the wall and cables, from the grounding bars connected to the drums of solvents. Take any material safety data sheet (MSDS) for a powder and look in section V; it highlights that any dry powder has the potential to attract and store a charge.
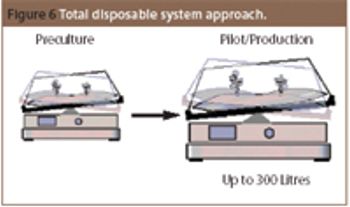
Over the past decade, disposable technologies have become a reality in biotech processes. The use of disposables in research and manufacturing allows high flexibility.

Brain and heart broth is still used in path labs, but, when it comes to biopharmaceutical manufacturing, cells are having to learn to do without animal components whenever possible.

Pfizer's restructuring plan provides yet another example of new supply-chain strategies by the pharmaceutical majors, which involve rationalization of manufacturing facilities and cost improvement. A review of these moves, an outlook for the pharmaceutical market in 2007, and analysis of US pharmaceutical production and trade.

Following the sales of its global branded pharmaceutical business, 3M stays the course in drug delivery as its Drug Delivery Systems Division advances technology in inhalation and transdermal drug delivery.

The US Department of Health and Human Services (HHS, Washington, DC) has awarded three vaccine makers a total of $132.5 million to advance their strategies for adjuvant-containing vaccines to combat the H5N1 strain of avian influenza. Under the contracts, each company will build capacity to produce either 150 million does of the vaccine or enough adjuvant for 150 million doses within six months after the onset of an influenza pandemic.

Darmstadt Germany (Jan. 8)-Merck KGaA closed on its roughly CHF 16.6 billion ($13.3 billion) deal to acquire a majority stake in the European biotechnology company Serono (Geneva, Switzerland), officially launched Merck Serono SA as a new entity within Merck KGaA, outlined its integration strategy, and announced plans to divest its generics business.
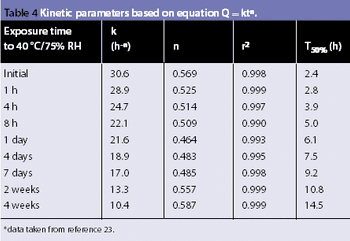
Water interacts with pharmaceutical solids at virtually all stages of manufacture, from synthesis of raw materials to the storage of the final dosage form. The interactions of water with powders is, therefore, a major factor in the formulation, processing and product performance of solid pharmaceutical dosage forms.

The concept of quality should not only apply to the manufacture of the media - it should also continue throughout the product's lifetime of usage.

There are currently several hundred biotech-based drugs in clinical trials, representing around a quarter of all drugs in development - a proportion that looks set to increase.

Biotechnology stocks dropped 14% in 2006, giving up more than half of the value they'd picked up during a "stellar" 2005, according to G. Steven Burrill, CEO of Burrill & Company (www.burrillandco.com), the venture and merhant banking company. "Overall, it wasn't a great year for biotech," said Burrill in a year-end analysis, indicating that the biotechnology industry will finish the year with its collective market capitalization essentially unchanged, at approximately $490 billion. As for the future, biotechnology will continue to fuel a the transformation in healthcare, a tansformation emphasizing earlier disease detection, more targeted treatments, and adjunctive support through enhanced nutrition. We will see further progress on the personalized, predictive, preventative front...with new products targeting the "individualization" of medicine in the marketplace. Other predictions for 2007 include...

Washington, DC (Dec. 13)-After considerable debate and negotiation, Congress this week passed four bills poised to affect pharmaceutical and biotechnology research, development, and manufacture. All are currently awaiting signature by the President.

Basel, Switzerland (Dec. 5)-Lonza Group Ltd. and Singapore?s Bio*One Capital have formed a joint venture, Lonza Biologics Tuas, to build a large-scale mammalian cell-culture facility in Singapore for as much as $350 million.

Brussels, Belgium (Dec. 5)-The debate over biosimilars in Europe was heightened last week between the European Generics Association and the International Alliance of Patients Organizations, a patient advocacy group that issued a briefing paper on biosimilars to the European Parliament, the legislative arm of the European Union (EU).
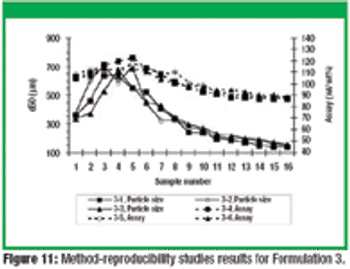
This article describes the design and development of a material-sparing fluidization segregation tester for use with pharmaceutical powders. This tester offers several improvements over the current ASTM standardized test practice. Less than 20 mL of material is required to characterize the fluidization segregation potential of a sample. Features of the tester include powder containment for potent compounds, in-process monitoring of the fluidization conditions, and sample retrieval without the need for subsampling or riffling for typical analyses.

Brand-name manufacturers seek to protect their products.

Makers of injectable drugs can increase their market share and stay competitive by devising a product life-cycle management strategy.
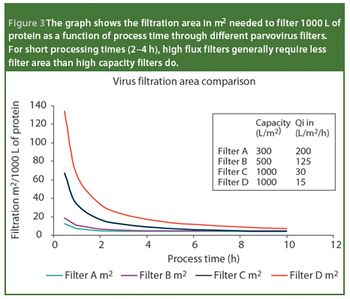
Virus safety of biotech- and plasma-derived therapeutics is ensured through complementary manufacturing and quality control measures that include the control and monitoring of raw materials, the validation and implementation of effective virus clearance technology and the monitoring of final filled product for the presence of virus. Virus filtration, which is considered a robust and effective virus clearance technology, is a common unit operation in the manufacture of biologicals. In this article, we review the points that must be considered when selecting a virus-retentive filter. The areas covered include regulatory considerations; selecting, optimizing and validating a virus filtration step; and process scale implementation - areas that are critical to users of virus filters.

Why do the biotech ventures mange to have such promising drug candidates while pharmaceutical R&D is facing a drug deficit?
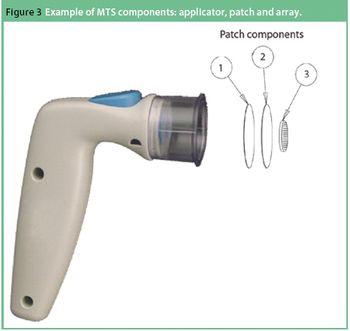
The needle and syringe have long been the standard delivery technology for vaccines. However, a confluence of market factors is driving new interest in alternative delivery systems that hold the potential to meet one or more of the following goals: improved antigen utilization, higher quality immune response, better stability and improved patient acceptance. Of particular interest are microneedle systems, otherwise referred to as microstuctured transdermal systems (MTS), that provide for targeted delivery of the vaccine formulation directly to antigen-presenting cells within the epidermis. This article provides a brief overview of MTS technology with an emphasis on solid-coated MTS for vaccine delivery.

Washington, DC (Nov. 20)-The US Department of Health and Human Services awarded contracts totaling $199.45 million to Sanofi Pasteur, Novartis, and GlaxoSmithKline PLC to manufacture 5.3 million 90-?g doses of influenza vaccine designed to protect against the H5N1 influenza virus strain.