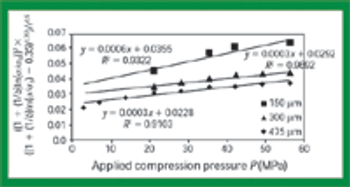
Quantitative data from the literature show strong relationships among average particle size, powder densification, tensile strength, and hardness.

Quantitative data from the literature show strong relationships among average particle size, powder densification, tensile strength, and hardness.
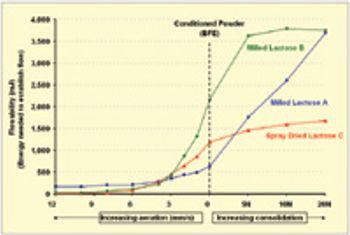
The pharmaceutical industry's focus on process understanding, monitoring, and control is driving manufacturers to take greater steps toward identifying possible manufacturing bottlenecks earlier in the development process. For tablet, capsule, and excipient producers, such efforts include taking a closer look at the flow-ability of their powders.
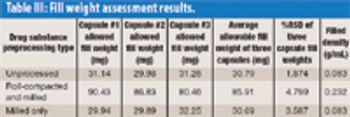
Using a novel automated microfilling system, the authors demonstrate that roller compaction followed by milling is a viable preprocessing technique for high-dose chemical-in-capsule dosage forms. The process results in higher bulk and tapped densities for drug substances compared with milling alone.

Though dissolution testing has been under scrutiny, it is still a powerful test method.
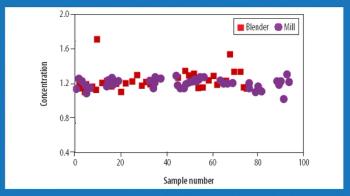
Almost all pharmaceutical manufacturing processes require handling and processing cohesive powders. The application of sufficient shear (i.e., the total deformation that the bulk of granular material undergoes under applied shear stress) is an essential factor in such processes. Sufficient shear is required to mill and de-lump materials, achieve sufficient flow, and homogenize cohesive ingredients. Shear mixing plays a critical role in the blending of dry powders, particularly for those that contain a minor cohesive component such as a solid lubricant or a drug. This mechanism is necessary to achieve a satisfactory homogeneity and disintegrate possible agglomerates. Excessive shear can be disadvantageous, however, and can lead to electrostatic buildup, attrition, and overlubrication.
Solid oral drug products are one of the oldest of all manufactured dosage forms (1). Today, the development of an appropriate formulation of drug and excipients and of an effective manufacturing process to create a tablet or capsule is slowly transforming from a practice of applied art to one of applied science. The US Food and Drug Administration supports this change by expecting sponsors of new drug applications to understand, describe, and control materials and processes as well as the risks associated with drug product manufacturing (2). These steps will ensure the consistent production of products that meet their specifications and remain safe and effective during their shelf life.

Control Development's (South Bend, IN, www.controldevelopment.com) blend uniformity and dryer monitor consists of a NIR spectrometer, a sampling head, computer, and software.

Generally, tablet and capsule film coatings are applied as aqueous or organic-based polymer solutions or dispersions, graduate student Sagarika Bose (University of Connecticut) explained during her Tuesday AAPS Graduate Student Symposium presentation, "Development and Evaluation of Solventless Photocurable Pharmaceutical Film Coating." However, organic film coatings can be flammable, toxic, and must comply with strict environmental regulations. Aqueous film coating can lead to the degradation of certain drugs by heat and water.
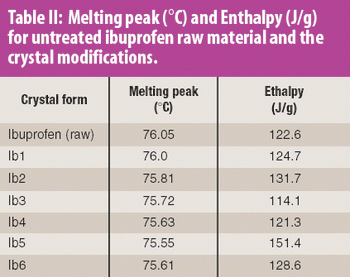
The common crystal form of ibuprofen was changed to optimize processing and manufacturing properties. Six modified crystal forms were prepared and assessed for dissolution, morphology, particle size, density, thermal characteristics, powder x-ray diffractometry, flow properties, and tabletability.

This review article discusses orally disintegrating tablets and their manufacturing technologies, development issues, and future trends.
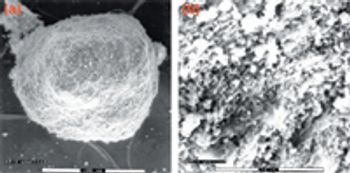
Although agitation improves drying efficiency and ensures uniformity of the final dry material, it can also affect the physical properties of the product as it dries. This study evaluates the effect of scale up and equipment selection on an active ingredient undergoing granulation during the drying process.

Pharmaceutical science and technology innovation
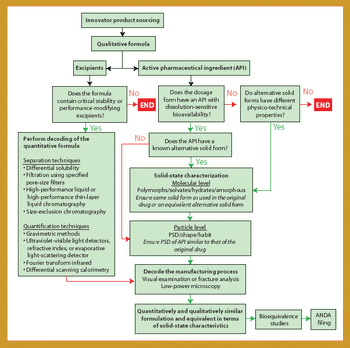
Being the first to gain the most is a fundamental principle in the generics business because several companies compete to create generics of successful products going off patent. For a generics company to maintain revenue growth in a market in which product prices continue to fall, it must secure a continuous flow of new products, with quality and speed to market being key drivers. Thus, generics companies must be highly skilled in product and process development (1), the generics business, and achieving bioequivalence-the most critical development area.

Self-Assembly Nanotechnology Improves Microencapsulation

Once considered mainly an afterthought in a company's lifecycle-management strategy, controlled-release dosage forms are now positioned at the forefront of many formulation strategies. In contrast to drug discovery, formulation work focuses not only on the intricacies of the active pharmaceutical ingredient (API), but also on fine-tuning the excipients, the release profile, and the delivery mechanism to provide optimal therapeutic benefit. Because of their wide range of applications and functionalities, especially in controlled-release therapies, polymers are among the most widely used excipients.

The principles of direct compression haven’t changed in more than a century. So why can the topic still pack a ballroom?
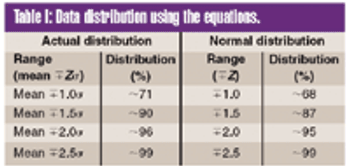
To meet the requirements of the USP ?755? Minimum Fill and ?698? Deliverable Volume tests, target fill levels greater than 100% must be established. This article proposes a criterion for establishing an appropriate target fill level such that a sample will have a 95% probability of passing these USP tests at 95% confidence.

Big Pharma Companies Team Up to Develop Once-Daily, Triple-Combination HIV Drug

New fixed-dose combination drugs aim to enhance safety and efficacy, while regulators clear a path for more drug–device combination products.

Albumin-Bound Nanoparticle Drug Nabs FDA Approval

The author reviews the effects of moisture on flow properties, tensile strength, Heckel plot, energies involved in compaction, and elastic recovery.

From solving complex formulation challenges to delivering full product-development programs, outsourcing services organizations provide valuable expertise, experience, and technical know-how.

With creative engineering and sophisticated chemistry, industry scientists offer big solutions for attaining small particle sizes and narrow distributions.

Freeze pelletization is a new and simple technique for producing spherical pellets for pharmaceutical use.
