
Trifarma cited for significant deviations in data collection and security, and employee training.

Trifarma cited for significant deviations in data collection and security, and employee training.

The pharma industry is moving toward commercial-scale continuous processes for small-molecule API manufacturing.

Transition metal-containing catalysts are widely used in organic synthesis, but many are based on expensive, rare metals. Researchers are seeking more sustainable alternatives.

Appropriate process design and engineering are critical for the production of small-molecule and biologic HPAPIs.

The Bulk Pharmaceuticals Task Force asks FDA to respond to concerns about reduction in API facility compliance inspections.
USP's Therapeutic Peptides Expert Panel discusses manufacturing processes and impurity control for synthetic peptide APIs.
Drug developers are taking a wait-and-see approach to the use of cocrystals.

The author examines the effect of compaction force, gap width, and sieve setup on granule size.
Increasing potency and growing interest in antibody-drug conjugates are creating challenges for manufacturers of HPAPIs.
The commercial availability of an increasing diversity of enzymes has led to the growing use of biocatalysts for API synthesis.

Recent regulatory initiatives designed to secure the global pharmaceutical supply chain will directly impact the global supply chain and API manufacturers.

BASF receives EXCiPACT Certification for the group of polyvinylpyrrolidone polymers, produced in Germany.

Avantor Performance Materials adds VWR International locations to its certified excipient distributor program.

CPhI-InformEx Report will to describe American pharma economy from European and domestic perspectives.

Emerging markets are an engine for growth in the custom-manufacturing and pharma markets.

PIC/S reviews deficiencies found during inspections of API manufacturing facilities, harmonizes GMP standards, and provides training for inspectors worldwide.

A review of this year's crop of the new molecular entities and new biologics license applications approved by FDA thus far in 2013.
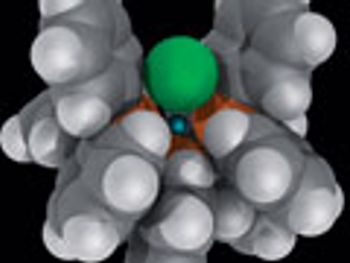
Fine-chemical companies, contract manufacturers, and researchers advance chemocatalysis and biocatalysis.

With the enforcement of the European Directive 2011/62/EU relating to medicinal products for human use regarding falsified medicinal products, new requirements are introduced for active substances. The new regulation requires among other things, documentation of the supply chain traceability.

Cedarburg Hauser Pharmaceuticals has upgraded its API plant in Wisconsin.

Pfizer advances its pipeline of antibody-based therapies using a host cell line that combines engineered glycosylation technology and enhanced gene-expression.

New partnership aims to eliminate months from the typical transition time required to move chemistry from the laboratory into commercial applications.
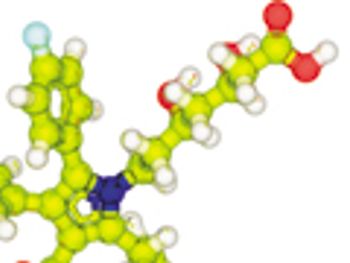
Developments involve stereoretentive cross-coupling, enantioselective alcohol silylation, strategies for amplifying signals in circular dichroism spectroscopy, and a synthetic route for the natural product ingenol.

Industry experts share their views on the outsourcing model and the current and future direction of contract chemical API manufacturing.
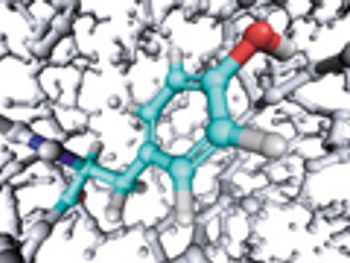
Commercial-scale amide formation and an improved process route for a tetracycline derivative are some recent developments in API synthesis.