
Company and People Notes: Also, Pfizer and Protalix enter agreement; Watson acquires Arrow Group; and more.

Company and People Notes: Also, Pfizer and Protalix enter agreement; Watson acquires Arrow Group; and more.

A joint biopharmaceutical manufacturing facility in India by Kenwell and Boehringer Ingelheim ushers in new era.

Kemwell (Bangalore, Karnataka, India) plans to build a new biopharmaceutical manufacturing plant in Bangalore, India, in a strategic collaboration with Boehringer Ingelheim (BI, Ingelheim, Germany), according to a Kemwell press release.

Strong growth in biopharmaceuticals bodes well for contract manufacturing, but the perils and the promises of pipelines remain.
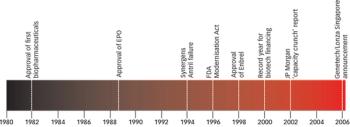
Biopharmaceuticals are the most rapidly growing segment of the pharmaceuticals market. Developing and marketing biopharmaceuticals are huge roles in almost every major pharmaceutical company's strategy. However, they are extremely complex molecules and are highly sensitive to the manufacturing processes used to produce them. These processes require exquisite control of living production systems, making, without a doubt, biopharmaceuticals one of the most challenging products of any type to manufacture.

A surge in capacity in contract microbial and mammalian cell-culture is underway to meet rising production needs for biopharmaceuticals.
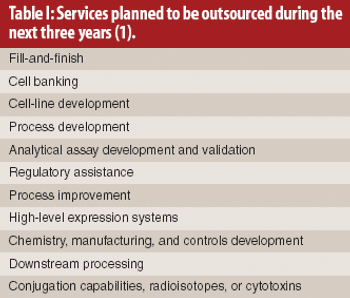
The authors analyze the supply–demand trends for contract biologics manufacturing and the strategies of pharmaceutical and biotechnology companies and their suppliers in the value chain.

Contract service providers should be concerned about the market perceptions of their performance.
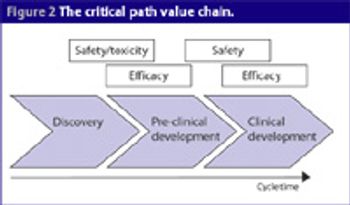
One of the biggest barriers research and academic institutions face is the ability to get discoveries made in the lab into clinical testing. Because only small amounts of drugs are used in these early studies, they represent fewer potential risks for people in these trials.

Is the lack of finance necessarily spelling doom for the European biotech industry?

A CMO in Singapore manufacturing commercial biopharmaceuticals is the result of a partnership between two biopharmaceutical companies.

Too many players and threats to the main customer base challenge European dose manufacturers.

Patheon is betting the whole is worth more than the sum of the parts in its acquisition of Puerto Rico contract manufacturer Mova.

The blockage in the new product pipeline may be breaking. CROs report increased demand for late-stage development services in their latest round of earnings reports.

Early development services continue to be a sweet spot for contract R&D services. The big question is how soon the impact of early-development candidates will be felt in late development and commercialization.

The contract services industry is fertile ground for the investment banking community this summer.

Data from the 2004 Outsourcing Survey indicate that pharmaceutical companies are spending more on outsourced drug development, but with fewer suppliers.

It's not clear whether contract service providers can deliver the performance improvements and costs savings that drug companies expect from large, long-term, semi-exclusive contracts.

Outsourcing is part of the public debate as politicians seek to explain the lag in job growth despite a recovering economy, and as the Bush administration presses its initiative to "competitively source" more agency activities.

December's Contract Services articles include "Using Virtual Private Networks to Gain Competitive Advantage," by Mark Tuomenoska and "Outsourcing Outlook," by Jim Miller.

November's Contract Services articles include "Outsourcing Xbox Manufacturing," by Heather Hayes, "Outsourcing Outlook," by Jim Miller, and "Quality in Pharmacovigilance," by Brian Edwards and Jeffrey Priem.

The European Union is attempting to reform its current business environment in hopes of restoring its role in the lobal R&D sector.

The author discusses the potential effect on CROs of pending major-pharma mergers and consolidations.

The author examines the concept of evaluating CRO operating performance in today's pharmaceutical industry.

The results of this 2002 survey of contract services decision makers reviews and forecasts the state of the outsourcing industry.