
Outsourcing activity remains strong and unlikely to abate, especially in more traditional areas.

Outsourcing activity remains strong and unlikely to abate, especially in more traditional areas.

CMOs may find opportunities in alternative expression services.
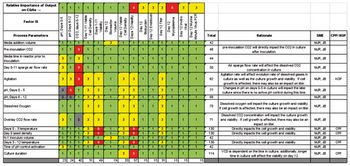
Traditional project decision-making is compared with a QbD approach.

Increased competition from CMOs in Asia means that Western CMOs need to understand fundamental changes in the market.

A changing biopharmaceutical industry is going beyond typically outsourced activities and is using CMOs for more challenging processes. Review the top 10 outsourcing trends.

With numerous biologics set to come off patent soon and the percentage of new therapeutics based on biomolecules growing, demand for contract manufacturing in the biopharma space is heating up.

Data from BioPlan Associates’ 10th Annual Report and Survey of Biopharmaceutical Manufacturing Capacity and Production suggest that the interest in disposable devices has begun to extend to biopharma operations beyond basic single-use bags and connectors.

Recipharm has invested EUR 32 million ($43 million) in its Wasserburg, Germany site to expand lyophilization capacity.

Advances in techniques and single-use systems are revolutionizing vaccine manufacturing.

Industry experts share their views on the outsourcing model and the current and future direction of contract chemical API manufacturing.

A CMO perspective on the CMO-client partnership.

Advances in techniques and single-use systems are revolutionizing vaccine manufacturing.
External manufacturing plays a crucial role in pharmaceutical companies’ supply strategy. The author examines market trends for the captive and merchant global market for active pharmaceutical ingredients and intermediates.
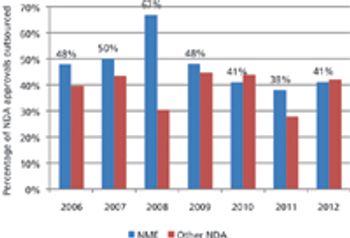
Approvals of new drugs are on an upward swing, but only a few CMOs are benefiting.

Is the contract-only CMO an endangered species?
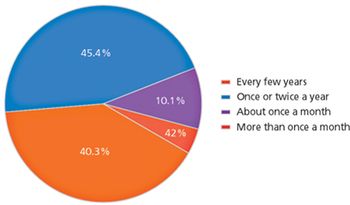
A UC Berkeley survey provides insight into biopharma's risk concerns and strategies.

Innovation resulting in improved productivity continues unabated and is a primary driver for many of the current biopharmaceutical trends.

Key talks from the recent PDA/FDA regulatory conference highlight room for improvement.
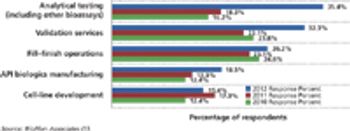
Budgets for biopharmaceutical activities are gaining in select functional areas except outsourcing.

Annual survey shows strong growth for service providers and promises to continue into 2013.

Jim Miller, president of PharmSource, examines the future direction of CROs/CMOs and the factors influencing the pharmaceutical contract services sectors.

Contract API manufacturers and fine-chemical producers roll out capacity and service expansions.

The evolving bio/pharmaceutical business model poses risk for CMOs.
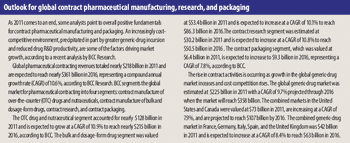
Expansion activity was limited as fine-chemical producers and CMOs of API and intermediates grapple with changing industry fundamentals.

In a quick-fire interview, Dr Stephen Taylor, Vice President & Commercial Director at Fujifilm Diosynth Biotechnologies, looks at some of the challenges facing biologics outsourcing.