
A Federal Circuit court ruled biosimilar makers must wait 180 days after receiving FDA approval before bringing drugs to market.

A Federal Circuit court ruled biosimilar makers must wait 180 days after receiving FDA approval before bringing drugs to market.

The assay will provide information on biosimilar comparability.

Novo Nordisk broke ground on an expanded production plant for insulin in Kalundborg, Denmark.

Teligent is expanding its manufacturing and R&D complex in New Jersey.
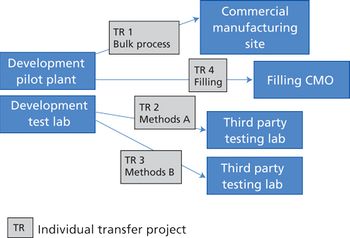
Integrating quality and compliance with technology transfer and careful project management are key in starting up a facility and launching a biologic drug.

Early planning for the integration of clean-in-place systems for equipment cleaning is key.

CPhI Pharma Awards seek nominations for excellence in development and manufacturing.

Polymers have played a key role as solubilizing excipients. Industry experts explain why polymer structures and functionalities are important considerations in formulation development.

Lipid-based drug delivery is increasingly being used to tackle oral bioavailability challenges resulting from poor solubility.

Aseptic spray drying provides an alternative to lyophilization as an enabling stabilization technology for parenteral biologic formulations.

FDA approved the Raplixa, the first spray-dried fibrin sealant, in May 2015 to help control bleeding in adults during surgery.

The Titan syringe pump from Syrris is a continuous-flow chemical processing module designed for lab, pilot, and production-scale applications.

A study by MilliporeSigma and the Economist Intelligence Unit reviews growth drivers and approaches to mitigate risk.

Antibodies to dengue may either confer protective immunity to the Zika virus or enhance disease severity in secondary, related infections such as Zika.

MilliporeSigma and the International Vaccine Institute in Seoul, South Korea aim to develop more robust, scalable vaccine manufacturing processes.

The monoclonal antibody for the treatment of two forms of multiple sclerosis has a target action date of December 28, 2016.

The agency recommends Zalmoxis, a new cell-based therapy to support stem cell transplantation in patients with high-risk blood cancer.

Studying broadly neutralizing antibodies in infants may lead to new pathways in HIV vaccine development.

A research team associated with Dr. Carl June announces it has discovered a way to engineer a patient’s own immune cells to recognize cancer-specific glycoantigens on tumor cells.

In a lawsuit against Celltrion, Janssen alleges the GE subsidiary infringed one of Janssen’s patents covering the method of making the cell-culture media that HyClone later supplied to Celltrion.

A new indication for Emergent BioSolutions’ BioThrax will give the drug market exclusivity through November 2022.

Pfizer broke ground at its Andover, Massachusetts campus on a clinical manufacturing facility for complex biologics and vaccines.

Hovione expands drug substance and HPAPI capacity in East Windsor, New Jersey.

A study published in BMJ indicates that rheumatic patients with anti-infliximab antibodies may have a similar cross reaction to infliximab biosimilars.

FDA approved Vaxchora intended for travelers who are at risk for the disease.