
Piramal Enterprises has entered into an agreement to acquire 100% stake in Ash Stevens all by cash for a consideration of $42.95 million plus an earn-out consideration capped at $10 million.

Piramal Enterprises has entered into an agreement to acquire 100% stake in Ash Stevens all by cash for a consideration of $42.95 million plus an earn-out consideration capped at $10 million.

Irvine Scientific’s new product range includes chemically-defined, serum-free media, to increase productivity of viral vectors and recombinant proteins in suspension cultures.

The agency has adopted guidelines on the pharmacovigilance of biological drugs.

Jacobs Engineering will provide engineering and construction services to expand the Novartis site in Huningue, France.

The GMP-certified facility will produce TruTag microtags for global pharmaceutical customers.

Zika vaccine development is hindered by technical challenges and funding shortfalls.

Topical semi-solids, like creams, lotions and ointments are comprised of a complex mixture of microstructures or colloidal phases, frequently referred to as the Q3. These microstructures may include micelles, lamellar phases, polymer matrices, liquid crystalline and crystalline states of wax excipients, as well as the solid states of active pharmaceutical ingredients. Different topical medicated semi-solid formulations, spanning a range of treatment indications, reveal a variety of different microstructures.

Researchers at the ARC Centre of Excellence in Convergent Bio-Nano Science (CBNS) in Melbourne, Australia have developed a drug-delivery technology that circumvents first pas metabolism in the liver, thereby, enabling higher drug levels to reach the systemic circulation.
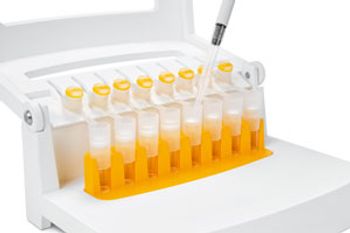
The Claristep filtration system from Sartorius improves preparation of samples for analytical testing.

Catalent Pharma Solutions teamed with Zumutor Biologics to develop antibodies with enhanced ADCC activity.

Jacobs Engineering Group was awarded a contract to provide engineering services and procurement for Alnylam Pharmaceuticals’ new manufacturing facility in Norton, Massachusetts.
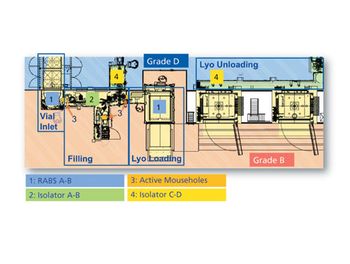
The design of Baxter BioPharma Solutions’ aseptic filling lines provides a case study in customizing containment systems for multi-product lines

Three-dimensional printing allows unique benefits to be built in to solid-dosage forms.

A technology management process identifies and evaluates new technologies in biopharmaceutical manufacturing to aid business decisions.

A Q&A with FDA to gain insight on FDA's views of three-dimensional printing and its regulation in drug manufacturing.
Industry experts discuss key considerations in the development of orally disintegrating tablets.

An MIT-developed system uses microbes for manufacturing small amounts of vaccines and other therapies.

Amgen discussed the drug-delivery approaches to two of its biologics in a recent second-quarter earnings call.

NIST’s monoclonal antibody reference material can be used as a standard for biopharmaceutical analytical quality control.

FDA and industry seek speedy Congressional approval of new user fee plan.

The Phase I trial will test Bavarian Nordic’s vaccine, which is manufactured using a vaccine vector based on smallpox.The recent resurgence of yellow fever incidences over the past six months has prompted health officials to ramp up the fight against the virus. Like the Zika virus, yellow fever is transmitted primarily through the bite of infected female Aedes aegypti mosquitoes.

The affinity purification company will set up shop at the former Merck & Co./GlycoFi facility in the Dartmouth Regional Technology Center in New Hampshire.

The project involves installation of a small-scale pressured agitated nutsche filter dryer in glass, integrated in a high-containment isolator to achieve an occupational exposure limit of less than 1 microgram per meter cube, 8-hour time weighted average.

The vaccine candidate has also won a Priority Medicines (PRIME) status from EMA.

Gamma-stable fluoropolymers are an alternative material for single-use bags and assemblies in biopharmaceutical manufacturing.