
Cold chain management is becoming pivotal to success in the pharmaceutical industry.

Cold chain management is becoming pivotal to success in the pharmaceutical industry.

By forming strategic collaborative partnerships with contract research organizations, pharmaceutical companies can take advantage of several strategies for accelerating drug development, maximizing profitability, and extending patent exclusivity.
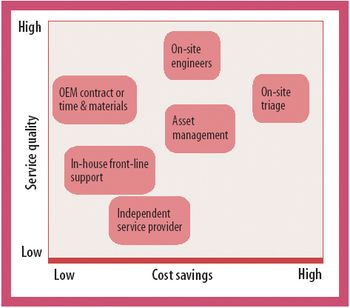
New maintenance models focused on efficiency improvements can deliver cost savings and enhanced service quality.
2005 has been a good year for the contract services industry, and 2006 promises to be nearly as successful, according to the 2005 PharmSource–Pharmaceutical Technology outsourcing survey. But as pharmaceutical outsourcing activity grows, companies are looking for more sophisticated ways to manage and control their contract services expenditures.

Protein Refolding Technology Improves Yields

Xanodyne Acquires aaiPharma Assets

Singapore is competing aggressively with India and China for a piece of the Asian sourcing business.

The Chapter 11 bankruptcy filing of aaiPharma (Wilmington, NC, www.aaipharma.com), parent of AAI Development Services, was the culmination of an agonizing year-long odyssey. It also was a first step in restoring one of the best-known brands in the CRO industry.

Compounding Pharmacies' Lawsuit Against FDA Will Continue

The unit-dose bar coding rule requires integrating many aspects of packaging design and control.

Major pharmaceutical companies are focusing more than ever on the prices CROs are charging for their services.

Crucell and NIH Sign Ebola Vaccine Manufacturing Contract

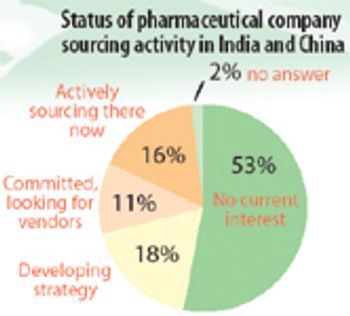
Achieving the promised benefits of offshore sourcing to India and China is more complicated than it seems.

How important will anticounterfeiting measures become to protect the European pharma industry?

Today's fast-paced global market place has created a critical need for efficient international supply systems in the healthcare sector...

After a five-year downturn, cautious optimism is taking hold among API manufacturers.

Novartis to Expand Generics Hold

Too many players and threats to the main customer base challenge European dose manufacturers.

From solving complex formulation challenges to delivering full product-development programs, outsourcing services organizations provide valuable expertise, experience, and technical know-how.

Patheon is betting the whole is worth more than the sum of the parts in its acquisition of Puerto Rico contract manufacturer Mova.

The blockage in the new product pipeline may be breaking. CROs report increased demand for late-stage development services in their latest round of earnings reports.


What should pharmaceutical companies make of recent corporate developments at several high-profile service providers?

… the real glue that holds the partnership - and project - together, even more so than a formal contract or quality agreement, is the ability to communicate regularly and honestly with each other.

A big problem for the industry seems to be that Phase II has become something of a 'black hole' for drug development; new drug candidates can't seem to escape once they enter.