
Plus, novel dosage forms and emerging trends.

Plus, novel dosage forms and emerging trends.
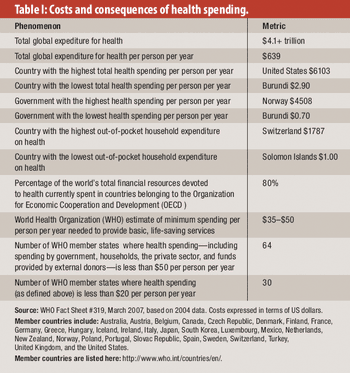
From healthcare to corruption to life expectancy, here's what we can learn from the past decade.

Directors and staff miss the mark when it comes to following procedures.

Pharmaceutical Technology Europe discusses some of the innovations in drug delivery that could shape the future of the pharma market.

There is a pressing need to improve the storage stability of currently available vaccines, and achieving this could facilitate mass vaccination campaigns and increase vaccination coverage on a global scale — particularly to underserved and remote regions of the world.

Tumour-penetrating microparticles (TPM), drug delivery vehicles that have been designed to specifically target and infiltrate peritoneal tumours, are more efficacious than standard chemotherapy, according to our preliminary research data.

BioNanomatrix (Philadelphia) names Edward Erickson president and CEO; and More.

Hospira, a specialty pharmaceutical company, has agreed to acquire Orchid Chemicals' Pharmaceuticals for $400 million.

Merck & Co. Acquires Avecia Biologics; Ambrilia Biopharma Closes Manufacturing Facility; And More.

Also, Abbott to acquire Starlims Technologies; GSK Biologicals to form alliance with Intercell.

Company and People Notes: Also, Pfizer and Protalix enter agreement; Watson acquires Arrow Group; and more.

Novartis officially inaugurated its large-scale flu cell-culture vaccine and adjuvant manufacturing facility in Holly Springs, North Carolina.

Also, Merck & Co. extends collaboration with Idera Pharmaceuticals; Pfizer establishes R&D center in China; more...
In this article, the authors evaluated the effects of the granulating binder level, binder type, water amount, and water-droplet size on the MADG process.

Readers can learn about the importance of measuring and controlling water activity in a comprehensive new book.

Particularly in a more connected world, individual contributions can make a difference.

Dumbed-down presentations and poor speaker selections are destroying a valuable industry tool.

With so many healthcare and pharmacy websites, consumers could use the agency's nod of support.

Leading experts share insight on polymorphism and crystallization.

Polymorph screening is a critical step in developing an active pharmaceutical ingredient for a pharmaceutical formulation.

The authors discuss a study that demonstrates the use of polyethylene oxide mixtures to investigate the effect of polymer viscosity on formulation robustness.

Generic-drug manufacturers look to expand into biologics and complex dosage forms. This article contains bonus online-exclusive material.

The application of roll compaction as a dry granulation method for three different drug types — herbal dry extract, poorly compactable drug and a sustained-release matrix tablet &#amp;151; was examined.

After further reviewing data on the H1N1 pandemic vaccines approved in Europe, the EMEA has reaffirmed their balance of benefits and risks in the context of the current H1N1 influenza pandemic. Additionally, the World Health Organization (WHO) has issued an update on the H1N1 situation in Europe.

Company and People Notes: Merck KGaA to build R&D hub in Beijing; Depomed appoints VP of operations; more...