
The new ready-to-use, packaging and production workflow solutions from DWK Life Sciences feature NextGen V and E-Z Ex-Traction vials to enhance security and minimize waste.

The new ready-to-use, packaging and production workflow solutions from DWK Life Sciences feature NextGen V and E-Z Ex-Traction vials to enhance security and minimize waste.

Cytena Bioprocess Solutions, a new Cytena subsidiary in Taiwan, will provide bioprocess solutions for pharmaceutical companies and research institutes.

Jubilant Cadista Pharmaceuticals is voluntarily recalling one lot of Drospirenone and Ethinyl Estradiol Tablets because of concerns regarding product efficacy.

The first generics of Lyrica have been approved by the FDA for neuropathic pain management and adjunctive therapy.
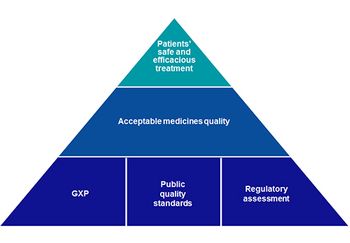
MHRA has launched a consultation to gain a greater understanding of the potential application of AQbD in pharmacopoeial standards and the future of medicines standards.

As drug supply chain security is of paramount importance, could radio-frequency identification solutions provide the optimal solution for pharma companies?

Novartis, Amgen, and Banner have announced their decision to discontinue the investigation into the BACE1 inhibitor CNP520 (umibecestat) as a preventative therapy for Alzheimer’s disease.

The Coalition for Epidemic Preparedness Innovations is partnering with Colorado State University to develop a single-dose vaccine candidate against Rift Valley Fever.

MHRA has issued a drug alert for the Emerade adrenaline auto-injector device, as a result of the device’s potential to fail in delivery of the dose due to blockage of the needle.

Development and manufacturing machine specialists, groninger, have opened a new showroom at its headquarters based in Crailsheim, Germany.

Catalent Biologics has entered into a long-term strategic agreement to develop and manufacture an AveXis gene therapy treatment for spinal muscular atrophy (SMA), Zolgensma.

Lonza plans to implement MES software to accelerate paperless, 24/7 production of drug capsules.

Process Automation Solutions becomes the latest partner to join the TrendMiner ecosystem.

Sartorius Stedim Biotech has unveiled a new single-use vessel for its ambr 250 modular benchtop automated mini bioreactor system that has been specifically designed for therapeutic cell lines.

The Bosch packaging machinery business, including the pharma unit, will remain intact in the transfer to private equity firm CVC.

Abcam has acquired the entire live cell line and lysates portfolio of gene-editing therapy developer, EdiGene.

Catalent recently held a groundbreaking ceremony at its Bloomington, Indiana pharmaceutical fill/finish site.

A July article in Science suggests that FDA enforcement activities have dropped significantly during the current US presidential administration.

Recent studies show that more companies are evaluating or using AI, and that top-down management support is a driving force.

The US Pharmacopeial Convention is partnering with the American Association of Colleges of Pharmacy to provide students with free access to quality standards.

cGMP deficiencies prompted FDA to withdraw approval for 31 Apotex ANDAs

Altaire is voluntarily recalling ophthalmic products because of concerns regarding quality assurance controls in the manufacturing facility.

FDA sent a warning letter to Ecometics, Inc. after the agency found CGMP violations at the company’s Norwalk facility.

The draft guidance describes how content should be organized in electronic submissions for all submission types under section 745A(a) of the FD&C Act.

Suheung’s new facility in Vietnam is dedicated to vegetarian capsule manufacturing.

An agreement with Sanofi gives Catalent access to commercial spray drying facility in Haverhill, UK.

Boehringer Ingelheim will combine assets from its cancer immunology portfolio with the KISIMA immunization platform from AMAL Therapeutics.

Gilead Sciences gains rights to clinical and preclinical programs in Galapagos’ portfolio through a 10-year global research and development collaboration.

A new starch manufacturing plant near Amsterdam, The Netherlands will give Colorcon customers a second source of supply.

Lubrizol’s purchase intravascular and nonvascular devices developer Bavaria Medizin Technologie, extends its scope of services.