
The standardization of upstream and downstream bioprocessing is growing, but several kinks need to be ironed out.

The standardization of upstream and downstream bioprocessing is growing, but several kinks need to be ironed out.

In a world where product recalls can mean the end of a company, all batches must be perfect.

Debottlenecking downstream mAb purification.

Experts discuss solutions for filter bacterial retention and related challenges. Contains online bonus material.

This column will address some of the questions on how single use systems are sterilized by gamma irradiation and what documentation may be requested by regulators.

New product reviews for January 2012.

Jerold Martin, senior vice president global technical affairs, at Pall Life Sciences, explains the importance and impact of single-use systems in sterile environments.

Contamination is almost always related to human error and there is a clear drive to reduce human implications in aseptic operations. This can be achieved in multiple ways.
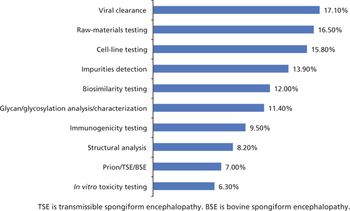
Biosimilar manufacturers need better expression systems and analytical tools to compete.

June and July saw three major US conferences on implementing single-use technologies: the IBC Single-use Applications meeting, the PDA Single-use Workshop and the Bio-Process Systems Alliance (BPSA) International Single-use Summit (ISUS). Jerold Martin highlights some of the key topics discussed at these meetings.
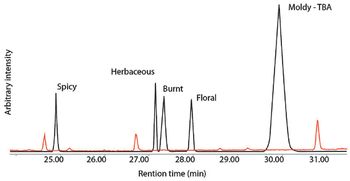
Combining olfactometry analysis with multidimensional gas chromatography–mass spectrometry provides an extremely useful analytical method for identifying aroma or odor notes from a sample.

The future, and increasingly the present, for aseptic operations is isolated robotics. Companies that wish to gain competitive advantages in their operations are stepping up and taking notice.

In a 2004 guidance, FDA says that the "use of redundant sterilizing filters should be considered in many cases." But not all manufacturers agree on what redundant filtration is.

Powder Systems Ltd's (PSL's) new GFD™ isolator combines the latest innovation to provide a high containment lab scale filtration and drying solution with the same reliability and benefits as PSL high containment production size filter dryers.
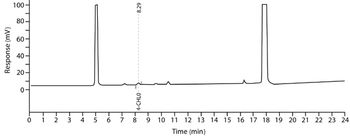
The authors provide an overview of methods for the quantitative determination of genotoxic impurities (GTIs) in active pharmaceutical ingredients.

Recently, there have been many innovations in the latest techniques and technologies in API purification. In particular, the trend to single-use systems has had a significant impact on processes.

Strategies to minimise particle levels in the finished product when using single-use technologies downstream of final filters.

A recent industry survey shows keen interest in improving bioreactors and cell-culture media.

Two popular methods for detecting protein aggregates are analytical ultracentrifugation (AUC) and size-exclusion chromatography?multiangle light scattering (SEC?MALS). These techniques? results correlate relatively well, but each one has its own strengths.
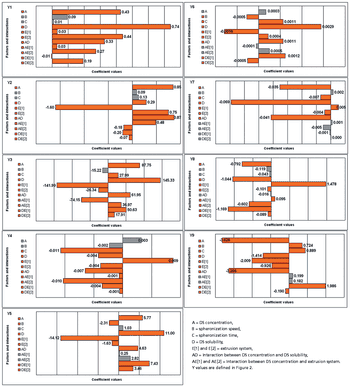
The authors compare three systems of single-screw extrusion using binary formulations for their suitability for producing pellets of various formulations and under various spheronization conditions.

Regions around the world are starting to follow the US trend in disposables and are working to solve common challenges.
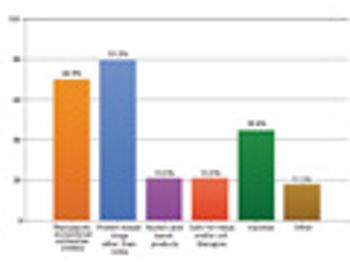
Results from our annual survey. This article contains online bonus material and is part of a special issue on Bioprocessing and Sterile Manufacturing.

We want to fill one of our oxygen-sensitive products inside an isolator system. Can we run an isolator with pure nitrogen and defined humidity?

Many facilities buy compressed gas tanks or evaporate liquid nitrogen to supply processes with dry, high-purity nitrogen. An in-house nitrogen generator, however, provides several significant benefits.

Over the past 10 years or so, we've seen single-use technologies explode from production-scale filter capsules, tubing and simple biocontainers to encompass sterile connectors, membrane chromatography adsorbers, bioreactors, mixers, and integrated platform systems with increasing levels of sensors and automated controls.