
In this episode fo the Drug Solutions Podcast, etherna’s vice-president of Technology and Innovation, Stefaan De Koker, discusses the merits and challenges of using mRNA as the foundation for therapeutics in oncology as well as for vaccines.

In this episode fo the Drug Solutions Podcast, etherna’s vice-president of Technology and Innovation, Stefaan De Koker, discusses the merits and challenges of using mRNA as the foundation for therapeutics in oncology as well as for vaccines.

Webinar Date/Time: Tue, Jun 11, 2024 10:00 AM EDT

CordenPharma’s partnership with GENEPEP expedites lead development and validation in project phases, and its LNP β-sitosterol supports sustainable emission reduction targets.
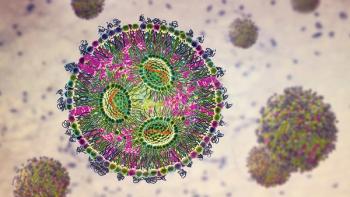
The LNP success in mRNA vaccines leads to their establishment as a standard in mRNA delivery.

In this exclusive Drug Digest video interview, Felicity Thomas, Europe/senior editor, Pharmaceutical Technology Group, interviews Dr. Krizia M. Karry, Global Head of Technical Marketing for BASF Pharma Solutions, on the topic of hot melt extrusion.

Charles River has entered into separate agreements with Axovia Therapeutics and Ship of Theseus to offer plasmid DNA manufacturing services for a gene therapy and a lead candidate program, respectively.

Webinar Date/Time: Fri, Apr 26, 2024 11:00 AM EDT

Webinar Date/Time: Wed, May 15, 2024 2:00 PM EDT

Genmab's acquisition of ProfoundBio will boost its clinical and preclinical pipeline of ADCs for targeted cancer treatment.

Advanced oral dosage forms allow for modulation of controlled- and sustained-release profiles.

Experts believe it is likely cancer cells are more common in the body than previously thought, but typically caught early and removed in the body’s cancer-immune cycle. When the system is in good working order (and protecting us), dead cancer cells release neoantigens.

FDA granted fast track designation to nipocalimab, which J&J is developing for helping alloimmunized pregnant adults treat FNAIT, a rare disease that poses risk to the fetus or newborn.

FDA has approved AstraZeneca's Ultomiris, marking the first and only long-acting C5 complement inhibitor for patients with NMOSD.

In this episode of the Drug Solutions Podcast, Jan Bekker, Vice President of Business Development, Commercial and Technical Operations at BioCina, discusses the latest analytical tools and their applications in the drug development market.

Webinar Date/Time: Tue, Apr 16, 2024 11:00 AM EDT

Webinar Date/Time: Thu, Apr 25, 2024 11:00 AM EDT

Webinar Date/Time: Wed, May 8, 2024 11:00 AM EDT

Mapp Biopharmaceutical will use ProBioGen’s GlymaxX technology for the development of an afucosylated antibody against Marburg virus infection

The draft guidance document provides support to sponsors with drugs in clinical development that treat the states of sporadic Alzheimer’s disease.

In a new manufacturing pact, Chime Biologics will produce DT-7012, Domain Therapeutics' antibody candidate for treating cancer that will soon be entering Phase I studies.

Webinar Date/Time: Wed, Apr 24, 2024 11:00 AM EDT

EMA has validated two MAAs submitted by AstraZeneca and Daiichi Sankyo for datopotamab deruxtecan in two types of cancer.

Johnson & Johnson’s $2 billion acquisition of Ambrx boosts its pipeline of next-generation ADCs for cancer treatment.

Webinar Date/Time: Tue, Mar 26, 2024 11:00 AM EDT

Under a global license and collaboration agreement, AbbVie and OSE Immunotherapeutics will aim to develop OSE-230, a mAb for treating chronic inflammation.