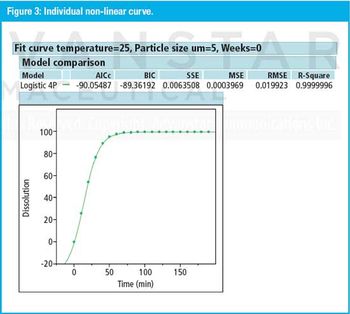
The right approach can provide a clear, statistically defendable method for determining dissolution and accelerated stability.

The right approach can provide a clear, statistically defendable method for determining dissolution and accelerated stability.
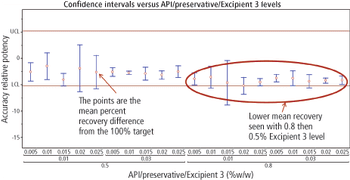
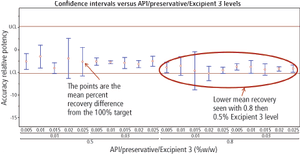
The authors describe a method-validation-by-design (MVbD) approach to validate a method over a range of formulations using both design-of-experiment and quality-by-design principles to define a design space that allows for formulation changes without revalidation.

A two-day workshop on the "science behind pharmaceutical stability" was held in conjunction with the Annual Meeting of American Association of Pharmaceutical Scientists (AAPS) on Oct. 21-22, 2011 in Washington, DC.

A two-day workshop on the "science behind pharmaceutical stability" was held in conjunction with the Annual Meeting of American Association of Pharmaceutical Scientists (AAPS) on Oct. 21-22, 2011 in Washington, DC.
IQ Consortium representatives explore and define common industry approaches and practices for applying GMPs in early development, with a focus on stability.
Published: April 2nd 2013 | Updated:

Published: November 2nd 2012 | Updated:

Published: November 3rd 2012 | Updated:
Published: September 2nd 2012 | Updated:
Published: July 2nd 2014 | Updated: