
Manufacturing
Latest News
Latest Videos

More News

Lonza signed a long-term supply agreement to manufacture CASGEVY (exagamglogene autotemcel) for Vertex at its facility in Geleen, the Netherlands, and plans to expand manufacturing to Portsmouth, NH, in the United States.

Hayley Crowe, executive vice-president and general manager Global Life Sciences at Ecolab discussed at BIO 2024 how technology innovations have enhanced downstream bioprocessing.

The drug manufacturing ecosystem Pharma 4.0, a term coined in 2017 by the International Society for Pharmaceutical Engineers, promises a revolution across drug discovery, manufacturing, and supply chain logistics, ensuring drugs are delivered precisely when needed, and improving overall patient care.

Lilly has added a $1 billion investment to increase biologics manufacturing at its Limerick, Ireland, site while unveiling its new $800 million facility in Kinsale.

Webinar Date/Time: Fri, Sep 27, 2024 11:00 AM EDT

During a presentation at the PDA/FDA Joint Regulatory Conference, Nicholas Violand of Johnson & Johnson and Paul Palmer of Paul R. Palmer Limited discussed how aging facilities can impact the quality of pharmaceutical products.

Greater collaboration and understanding are needed to ensure the correct grade of excipient is used in drug products.

Webinar Date/Time: Wed, Oct 2, 2024 9:00 AM EDT

New technologies improve efficiency and remove operator intervention.

Viral vectors and other complex biologic modalities require more specificity and higher sensitivity to detect and distinguish contaminants.

Rentschler Biopharma has expanded its service offerings at its Stevenage, UK, site with a new lentiviral vector manufacturing toolbox.
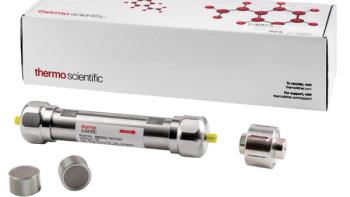
Thermo Scientific’s columns offer fast run times, optimal separation efficiency, and reliable results.
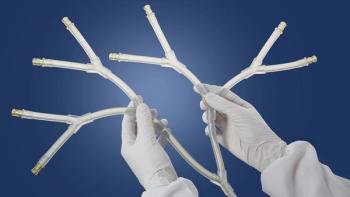
Freudenberg Medical’s new offerings include single-use Y-connector manifolds, tubing assemblies, and bottlecap assemblies, among others.

Charles Ross & Son Company has introduced the ROSS CDA-300 Dual Shaft Mixer, consisting of two agitators and a disperse with an open disc blade.

Asahi Kasei Medical now offers its Planova virus removal filters for use in biopharma manufacturing and plasma derivatives manufacturing.

The approach makes use of clinical mass spectrometers that are already available in hospitals around the world.

This is the company’s largest single investment in the Sweden Biomanufacturing Center, in Södertälje, which opened in 2021.

Vernal Biosciences’ CEO and founder, Christian Cobaugh, discusses the outsourcing landscape for mRNA production.

Genentech will have access to Sangamo’s capsid delivery platform and epigenetic regulation capabilities to develop genomic medicines for certain neurodegenerative diseases.

Webinar Date/Time: Tue, Sep 17, 2024 9:00 AM EDT

Webinar Date/Time: Wed, Sep 18, 2024 11:00 AM EDT

The growing use of continuous manufacturing in the pharmaceutical industry merits a review of the application of dry cleaning and sanitization methods, especially for non-sterile oral solid drug product manufacturing of dry powders.

With the agreement, Andelyin Biosciences will add MyoAAV plasmids developed by Broad Institute of MIT and Harvard to its AAV Curator Platform.

US Specialty Formulations plans to invest $15 million to expand biopharma operations at its Allentown, Pa.,facility.
Platform processes and effective risk assessments help overcome time and cost challenges.
















