Conference Coverage
Trending on Pharm Tech
Drug Digest: Outsourcing Partnerships Fuel Global Biopharma Discovery and Scale-Up
The Role of Standards in Sustainability
FDA Grants Priority Review For Pfizer’s Marstacimab for Hemophilia A or B
Shaping the Future of Microbial Development: From Gene to GMP
Women in STEM: Cultivating Scientific Confidence
Latest
Shorts










Drug Digest
Drug Digest: Outsourcing Partnerships Fuel Global Biopharma Discovery and Scale-Up
Strategic outsourcing utilizes external expertise to accelerate the biopharma pipeline, integrating specialized workflows that drive efficiency from discovery through early clinical trials.
Drug Digest: Closing the Gap in Modern Process Control and Automation for QC/QA
Biopharma quality control is being revolutionized by advanced analytics and digital transformation through a synergy that boosts quality and scalability.

Drug Digest: Strategic Alliances and Technology Integration in the Evolving Biopharma Landscape
In this episode of Drug Digest, the discussion centers around the optimization of biopharmaceutical manufacturing and supply chain efficiency through the mastering of AI capabilities, data integration, and critical validation challenges.
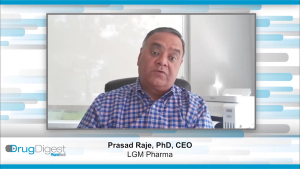
Drug Digest: Small Molecules, APIs, and Excipients—Trends, Challenges, and Opportunities
In this exclusive Drug Digest video, Steve Barr from SK pharmteco and Prasad Raje from LGM Pharma explore how supply chain pressures, sustainability, and AI adoption are reshaping small molecule development and excipient use in pharma.

Drug Digest: Speed, Flexibility Top Large-Molecule Manufacturing Trends
In this exclusive Drug Digest video, Deepak Bahl from Roquette and Jagruti Patel from Lonza look at strategies for accelerating early-stage development while reducing risk, approaches to speeding up timelines for complex formats without sacrificing quality, and maintaining a flexible CMC process that ensures quality.

Drug Digest: Patient Preference Drives Solid Dosage Trends
In this exclusive Drug Digest video, Adi Kaushal from Lonza summarizes the current state of the oral solid dosage market, identifies the main challenges to drug developers, explains how CDMOs are leveraging expert techniques to improve OSD bioavailability, and looks into the future of advancements in drug delivery.
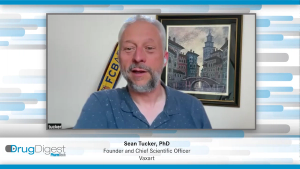
Drug Digest: Keeping Pace with Progressive Technologies in Biopharma Development
This episode of Drug Digest will discuss where the significant bottlenecks are in the development and manufacturing of new molecules resulting from progressive technologies, such as AI and ML, and how best to approach these challenges.
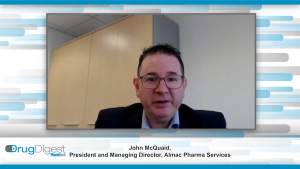
Drug Digest: Strategic Partnerships
In this exclusive Drug Digest video interview John McQuaid from Almac Pharma Services and Sridevi Khambhampaty from Shilpa Biologics delve into the evolution of the bio/pharma outsourcing market and look at how service-providers’ strategies have adapted to meet demand.
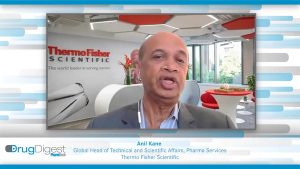
Drug Digest: Advances in Small-Molecule Manufacturing
In this exclusive Drug Digest video interview Anil Kane from Thermo Fisher Scientific will be tackling the topic of advances in small-molecule manufacturing and several other experts will provide brief commentaries on associated topics.

Drug Digest: Challenges and Triumphs in Next-Generation Biotherapeutic Development
In this episode of Drug Digest, industry experts discuss their experiences in dealing with the challenges of developing and manufacturing next-generation biotherapeutics as well as triumphs that move the industry as a whole forward.
Videos
All News

In part two of this four-part series regarding rare diseases, the author examines how economic and structural forces are driving an access gap.

ACG’s Shirwal site is the first pharmaceutical packaging site in the WEF Lighthouse Network, using digital tools to cut defects and boost sustainability.

PharmTech spoke with Elisabeth Gardiner, chief scientific officer at Tevard Biosciences, about how hands-on, early exposure to STEM fields can demystify complex concepts.

Cellares is partnering with Stanford Medicine to automate gene-edited stem cell therapy for HIV and more than 19 rare diseases.

FDA has granted priority review to AstraZeneca's Datroway for the first-line treatment of metastatic TNBC treatment.
Walid Kamoun, PhD, and Alex Philippidis discuss AI’s Power to Lower Costs while Elucidating Tumor Progression, NVIDIA and Eli Lily’s Deal, and Revolution Medicine’s lack of a Deal.

PharmTech spoke with Dr. Stacy Lindborg, president and CEO of IMUNON, about her experience at IMUNON, which is developing a novel DNA-based approach to treat ovarian cancer.
The companies’ “Change the Target. Change What’s Possible” targets the potential of Factor XIa inhibition in the development of treatments for thromboembolic disease.

Anthony Lakavage, executive vice president and head of Global External Affairs at USP, talks about how sustainability efforts are more than just protecting the environment and how pharmaceopeias are participating in the endeavor.
EMA recommends Kygevvi for rare thymidine kinase 2 deficiency. It is the first authorized treatment for this disease.

The EMA recommends a GLP-1 for MASH, a couple rare disease medications, and a flu vaccine among six new medicine and nine extension recommendations.

EMA recommends Rezurock (belumosudil) for chronic graft-versus-host disease, with the drug. Meetings unmet needs via conditional authorization.

The PreCheck Pilot Program is designed to strengthen the US pharmaceutical supply chain by increasing regulatory predictability and streamlining the assessment of new domestic manufacturing sites.

PharmTech spoke with Katy MacLellan, Technical Team leader, Symbiosis Pharmaceutical Services Ltd., about the women in the pharmaceutical industry that inspire her.

This week's coverage discusses the M&A report, $50B+ investments, automated cell therapy, vaccine approvals, and risks from the US leaving the WHO.

In a 2026 industry outlook interview, Raj Puri explains balancing speed with the financial and regulatory risks of accelerated drug launches via alignment.

M&A report claims that pharma is now targeting vertical integration and manufacturing control in radiopharma, GLP-1s, and ADCs to secure the supply chain.

Eli Lilly will invest $3.5 billion in a Pennsylvania facility for injectable weight-loss medicines, creating 850 new jobs and starting by 2031.

Vetter's €1.5 billion expansion adds German commercial and Illinois clinical sites to support global sterile injectable drug supply.

In a 2026 industry outlook interview, Raj Puri discusses how the tariffs hinder US pharma investment but drive demand for geographically segregated supply chains.

The ROSS CDA-200 optimizes viscous pharmaceutical batches via independent agitation, vacuum, and thermal control.

Cellares raises $257M to scale its automated manufacturing model and validate a move toward becoming an integrated development and manufacturing organization, or IDMO.

Repertoire’s T cell receptor-epitope discovery platform will be used to develop tolerizing therapy development candidates with Eli Lilly and Company.

Halo Pharma becomes a standalone drug product CDMO after the sale of Noramco’s API assets, adding sterile services by H2 2026.

With a potential US government shutdown looming, Megha Sinha, Kamet Consulting, says to treat FDA disruptions as predictable risks by protecting milestones and early readiness.
In part one of this four-part series regarding rare diseases, the author explores how rare disease treatment development evolved from a niche area to a cornerstone of biopharma growth.

With a potential US government shutdown looming, Megha Sinha, Kamet Consulting, discusses the impacts on pharma manufacturing and development.

Visit PharmTech.com for upcoming coverage of the International Day of Women and Girls in Science on February 11.

WuXi Biologics and HanchorBio partner to accelerate multi-functional fusion protein translation via end-to-end CMC and GMP services.

Laine Mello, director of Marketin, Ecolab, details how AI, continuous manufacturing, and Pharma 4.0 drive bioprocessing efficiency and cost savings.

US WHO withdrawal triggers pharma regulatory and supply chain risks while impacting global R&D collaborations.

Raj Puri, CCO, Argonaut Manufactoring, provides key insight on navigating tariffs, supply redundancy, and the risks of compressed timelines.
The European Commission has approved GSK’s adjuvanted recombinant respiratory syncytial virus vaccine, Arexvy, for use in adults aged 18 years and older.