
Current good manufacturing practices for oral solid dosage forms protect the product from contamination and potential errors.

Current good manufacturing practices for oral solid dosage forms protect the product from contamination and potential errors.

What impact will EMA’s decision to phase out COVID-19 regulatory flexibilities have on the pharma industry?

Manufacturer lawsuits prompt CMS to [slightly] modify pricing plan.

An intelligent drug development paradigm can enable small pharma to implement an effective early drug development approach.

The program aims to aid submissions from external stakeholders and FDA staff.

The guidance document outlines recommendations to limit potential carcinogenic risk.

In this episode, David Tisi and Reenal Gandhi provide expert insights into advances and innovations changing the drug dosage form landscape.

Pandemic, PBM, and other policies on hold during the summer recess.

Tarsus Pharmaceuticals’ Xdemvy (lotilaner ophthalmic solutions) is designed to treat Demodex blepharitis by directly targeting Demodex mites.

The damage to the facility, which produces 8% of US injectable drug consumption, should not significantly affect the current supply.

The framework is now expanded beyond COVID-19 vaccines and treatments.

The paper highlights the potential of AI in the medicinal product lifecycle and the approach that developers should take with it.

These recommendations will keep key antibiotics for respiratory infections available when they are needed most.

The document’s recommendations outline what studies and reports are required for changes to cell and gene therapy manufacturing.

The new drug may help vulnerable children resist RSV in the coming fall and winter season.

Laboratoire HRA Pharma norgestrel is the first daily tablet for nonprescription use to prevent pregnancy.

Eisai’s lecanemab-irmb was converted from an accelerated approval to a traditional approval.

The agency is reviewing the medicines after receiving reports of self-injury and suicidal thoughts in patients using the type 2 diabetes treatments.

EMA looks to focus on mRNA vaccines because their classification depends on the target and/or whether they are obtained chemically or biologically.

FDA's approval of Leqembi paves the way for wider coverage of the drug by Medicare and establishes a process for further clinical testing and evaluation of treatments for this widespread, debilitating condition

EMA, the European Commission, and HMAs are phasing out the flexible regulations put into place during the pandemic.

Full Marketing Authorization has been granted for Novavax’s COVID-19 vaccine by the European Commission.

FDA has approved Roctavian (valoctocogene roxaparvovec-rvox), a gene therapy from BioMarin Pharmaceutical for treating severe hemophilia A in adults.

The degree of formality in a risk management process should be customized to the organization’s particular needs and the risks involved.
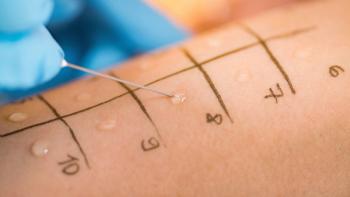
FDA’s new guidance on in-vitro permeation test studies published in October 2022 gives technical and statistical requirements for conducting these tests to compare topical generic drugs with their reference products.